
Structure-Based Discovery of SD-36 as a Potent, Selective, and Efficacious PROTAC Degrader of STAT3 Protein | Journal of Medicinal Chemistry
Efficacy, tolerability, and safety of an innovative medical device for improving oral accessibility during oral examination in special-needs patients: A multicentric clinical trial | PLOS ONE

Abbreviations: CR: Complete response; PR: Partial response; SD: Stable... | Download Scientific Diagram
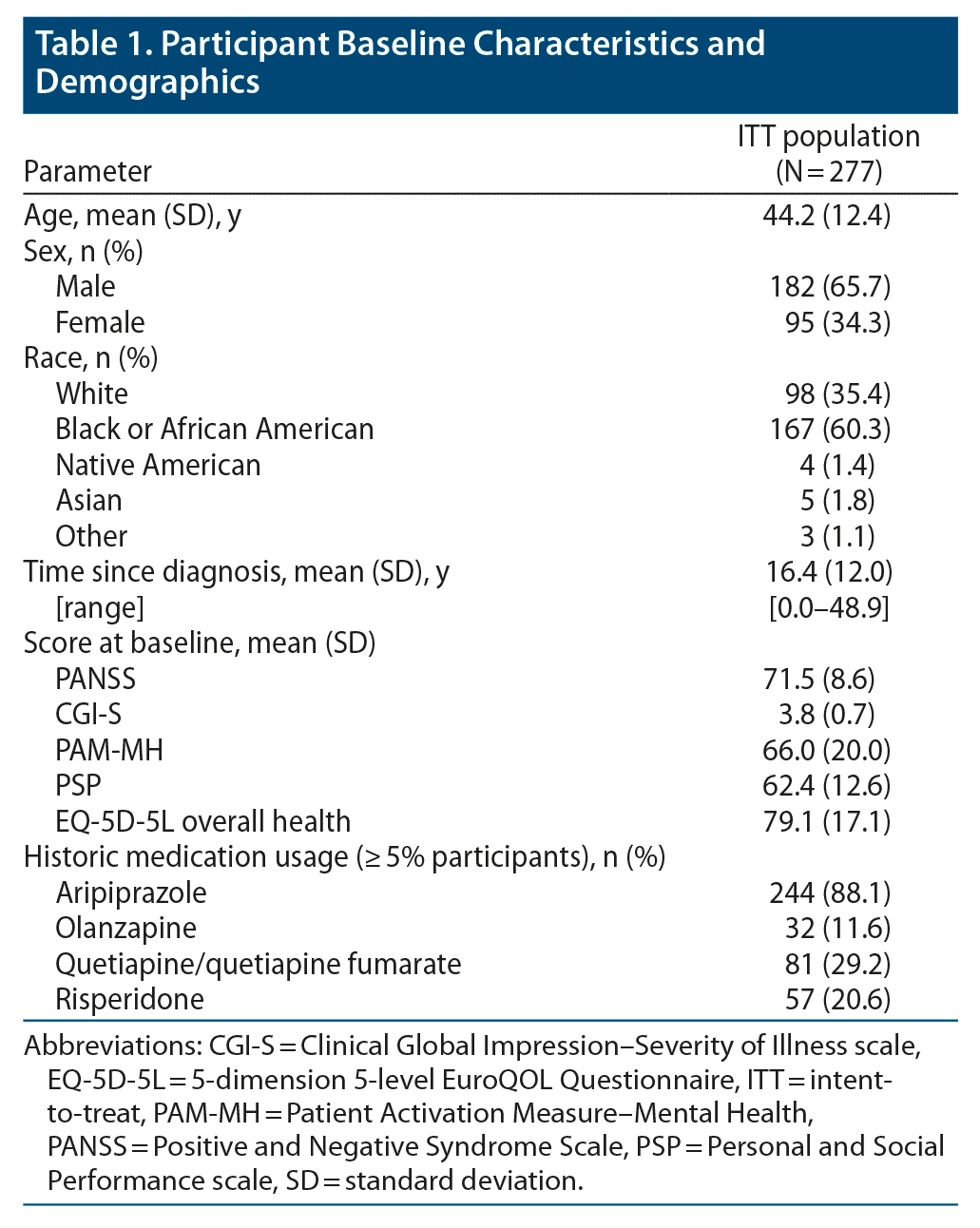
Phase 3b Multicenter, Prospective, Open-label Trial to Evaluate the Effects of a Digital Medicine System on Inpatient Psychiatric Hospitalization Rates for Adults With Schizophrenia | Psychiatrist.com

Abbreviations: CR: Complete response; PR: Partial response; SD: Stable... | Download Scientific Diagram

A Phase II, Open-Label Clinical Trial of Intranasal Ketamine for Depression in Patients with Cancer Receiving Palliative Care (INKeD-PC Study) - Inergency
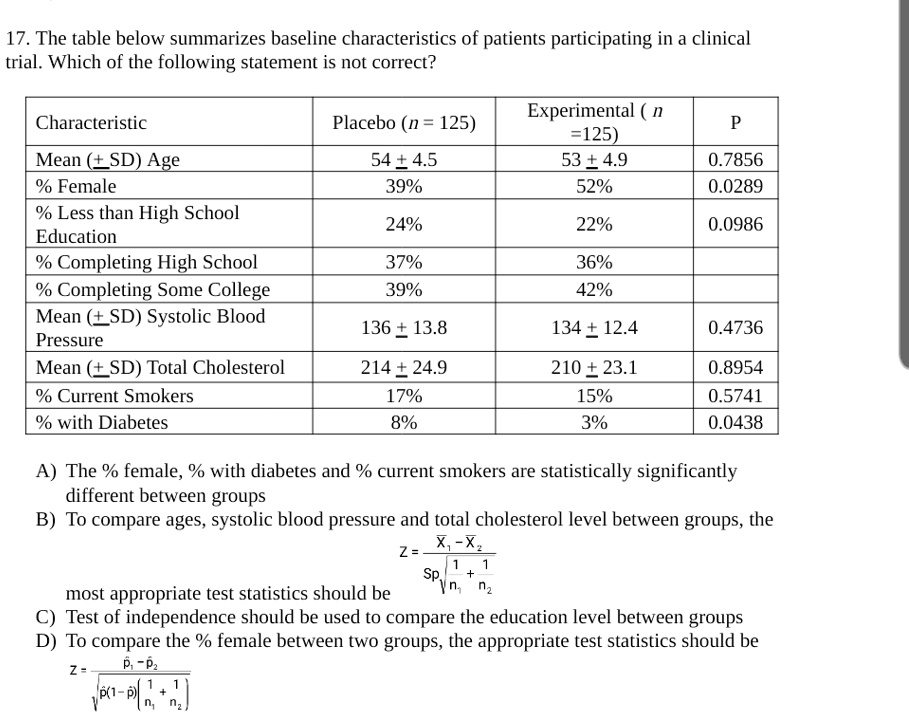
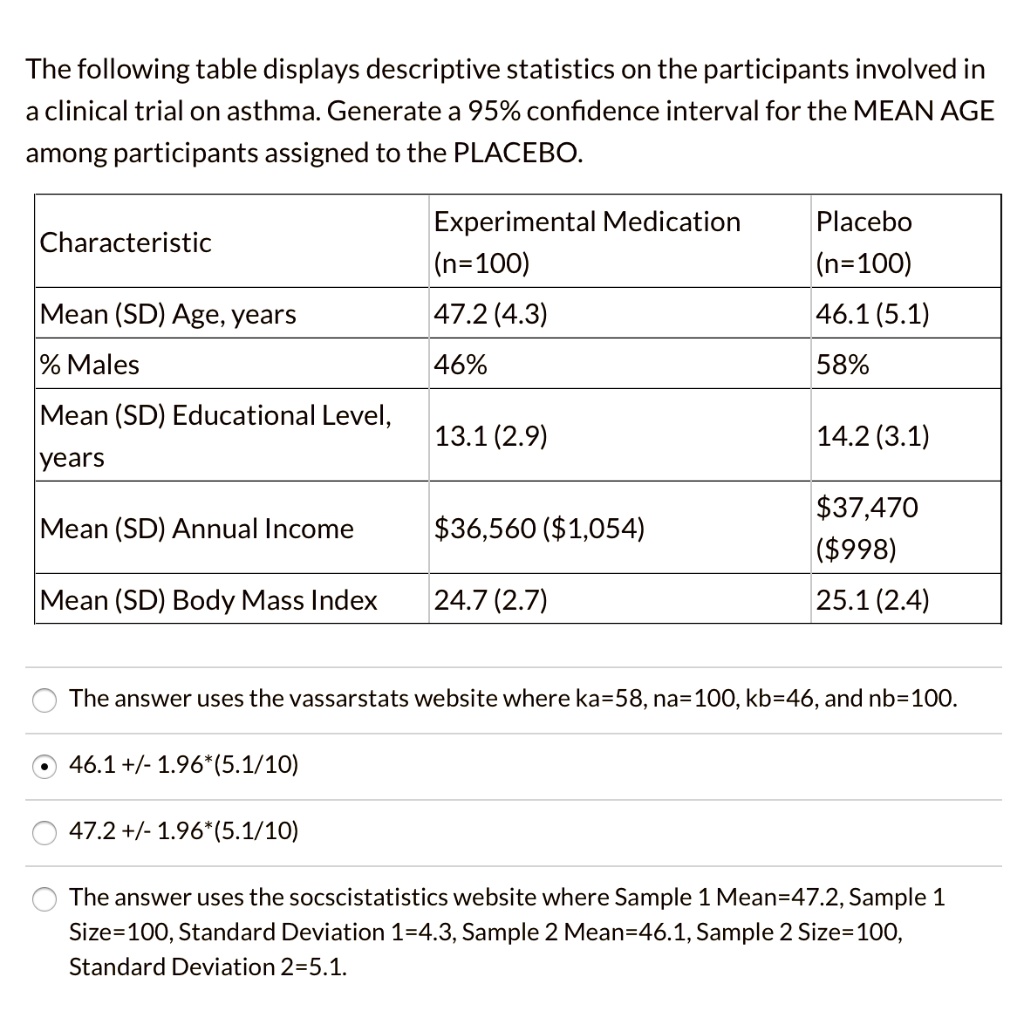
SOLVED: 17. The table below summarizes baseline characteristics of patients participating in a clinical trial. Which of the following statement is not correct? Experimental =125) 53+4.9 52% Characteristic Placebo (n = 125)
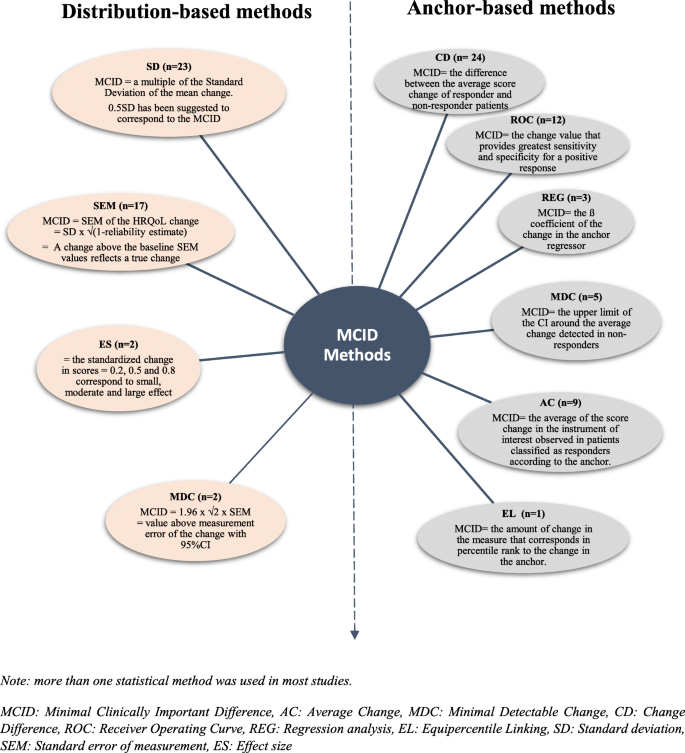
How is the minimal clinically important difference established in health-related quality of life instruments? Review of anchors and methods | Health and Quality of Life Outcomes | Full Text
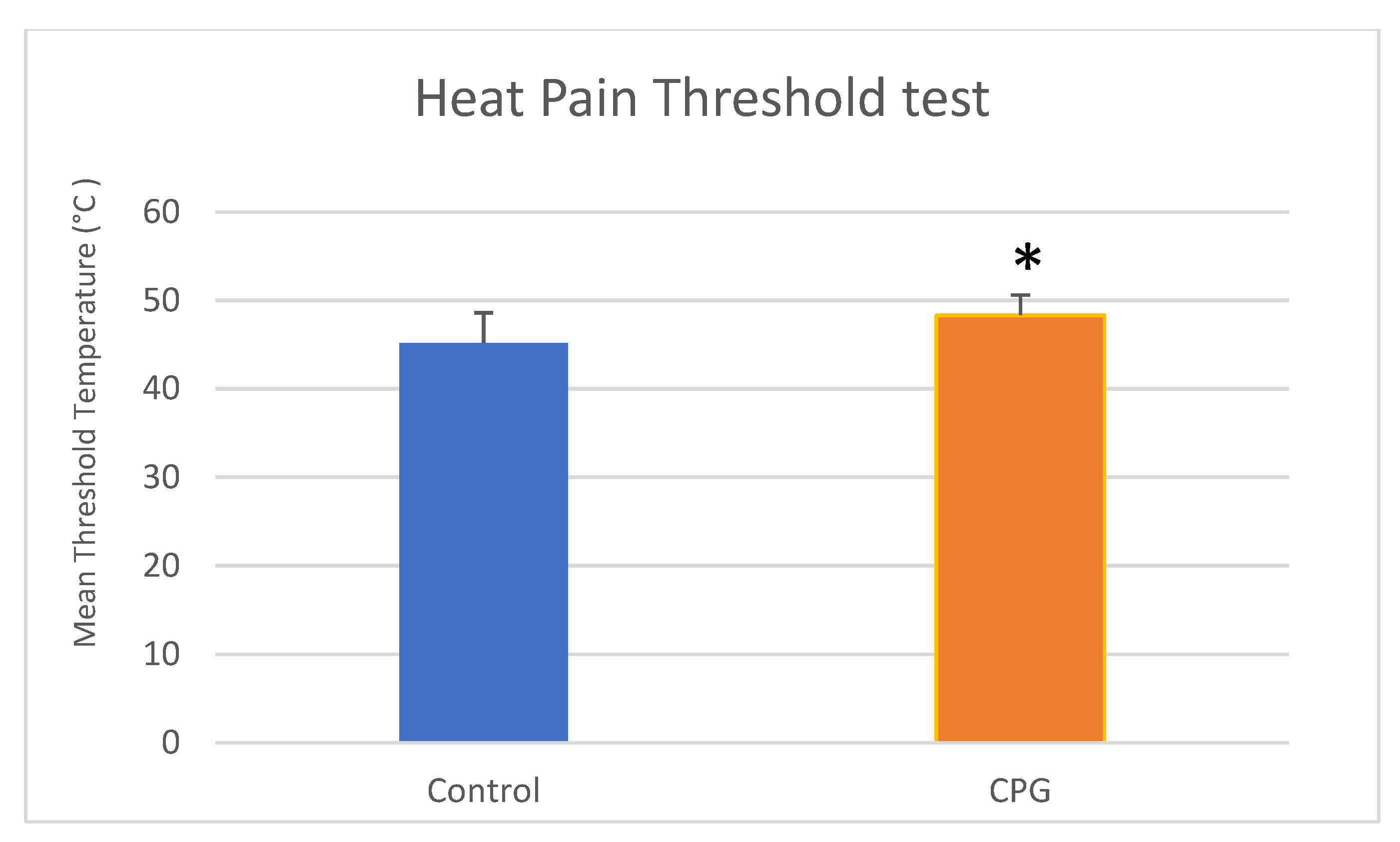
Biomedicines | Free Full-Text | Outcomes of a QST Protocol in Healthy Subjects and Chronic Pain Patients: A Controlled Clinical Trial

How Many Cancer Clinical Trials Can a Clinical Research Coordinator Manage? The Clinical Research Coordinator Workload Assessment Tool | JCO Oncology Practice

Clinical effectiveness and cost-effectiveness of tailored intensive liaison between primary and secondary care to identify individuals at risk of a first psychotic illness (the LEGs study): a cluster-randomised controlled trial - The






:max_bytes(150000):strip_icc()/recist-5206400_final-04c81c00d8cc4583975603ca53a6ff98.jpg)
